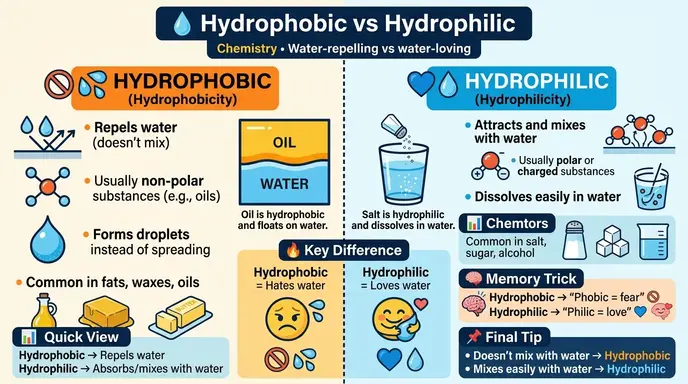
The difference between hydrophobic and hydrophilic is that hydrophobic substances repel water, while hydrophilic substances attract and interact with water.
Many learners get confused about the difference between hydrophobic and hydrophilic because both terms describe how materials behave with water at a molecular level.
If you’ve ever wondered what is hydrophobic vs hydrophilic, how they work, or why they matter in real life, this guide will clearly explain the difference between hydrophobic and hydrophilic in a simple and practical way.
⚡ Quick Answer: Difference Between Hydrophobic and Hydrophilic
- Hydrophobic: Repels water
- Hydrophilic: Attracts water
👉 Example: Oil is hydrophobic, salt is hydrophilic.
📖 Definition of Difference Between Hydrophobic and Hydrophilic
- Hydrophobic: A substance that does not mix with or absorb water because it repels water molecules.
- Hydrophilic: A substance that easily mixes with or dissolves in water due to its attraction to water molecules.
🔊 Pronunciation (US & UK)
- Hydrophobic: UK /ˌhaɪ.drəˈfəʊ.bɪk/ | US /ˌhaɪ.drəˈfoʊ.bɪk/
- Hydrophilic: UK /ˌhaɪ.drəˈfɪl.ɪk/ | US /ˌhaɪ.drəˈfɪl.ɪk/
Now let’s understand this clearly.
📊 Comparison Table: Hydrophobic vs Hydrophilic
| Feature | Hydrophobic | Hydrophilic | Explanation |
| Water Interaction | Repels water | Attracts water | Core difference |
| Solubility | Insoluble in water | Soluble in water | Behavior |
| Molecular Nature | Non-polar | Polar/charged | Chemistry |
| Examples | Oil, wax | Salt, sugar | Common items |
| Surface Behavior | Water beads up | Water spreads | Visual effect |
| Biological Role | Cell membranes (lipids) | Blood, cytoplasm | Function |
| Usage | Waterproof materials | Cleaning products | Application |
| Bonding | Weak with water | Strong with water | Interaction |
🔍 KEY DIFFERENCES EXPLAINED BETWEEN HYDROPHOBIC AND HYDROPHILIC
1️⃣ Interaction with Water
Hydrophobic repels water, hydrophilic attracts it.
👉 Example: Oil floats on water
2️⃣ Solubility
Hydrophilic substances dissolve, hydrophobic do not.
👉 Example: Salt dissolves easily
3️⃣ Molecular Structure
Hydrophobic molecules are non-polar, hydrophilic are polar.
👉 Example: Charge difference matters
4️⃣ Surface Behavior
Hydrophobic surfaces cause water droplets, hydrophilic surfaces spread water.
👉 Example: Waterproof vs absorbent
5️⃣ Role in Biology
Hydrophobic parts form membranes, hydrophilic parts interact with fluids.
👉 Example: Cell structure
6️⃣ Practical Uses
Hydrophobic used in coatings, hydrophilic in detergents.
👉 Example: Real-world applications
💡 What Is the Difference Between Hydrophobic and Hydrophilic in Simple Words?
In simple words, hydrophobic means “water-fearing”, while hydrophilic means “water-loving.”
👉 Repel vs attract water.
🧠 Why Do Hydrophobic or Hydrophilic Properties Exist?
They exist because of how molecules interact with water:
- Hydrophobic molecules avoid water due to lack of charge
- Hydrophilic molecules bond with water due to polarity
👉 Chemistry controls behavior.
🌍 Difference Between Hydrophobic and Hydrophilic in Real Life
In real scenarios:
- Raincoats are hydrophobic
- Towels are hydrophilic
👉 Everyday materials show this difference.
⚖️ DIFFERENCE + SIMILARITY TABLE OF Hydrophilic and Hydrophobic
| Feature | Hydrophobic | Hydrophilic | Similarity |
| Type | Chemical property | Chemical property | Same concept |
| Function | Interaction with water | Interaction with water | Water-related |
| Field | Chemistry | Chemistry | Scientific use |
| Application | Materials | Materials | Practical use |
| Behavior | Molecular | Molecular | Based on structure |
| Role | Natural systems | Natural systems | Biological relevance |
This table clearly shows the difference and similarity between difference between hydrophobic and hydrophilic for quick understanding.
❌ Common Mistakes with Hydrophobic or Hydrophilic
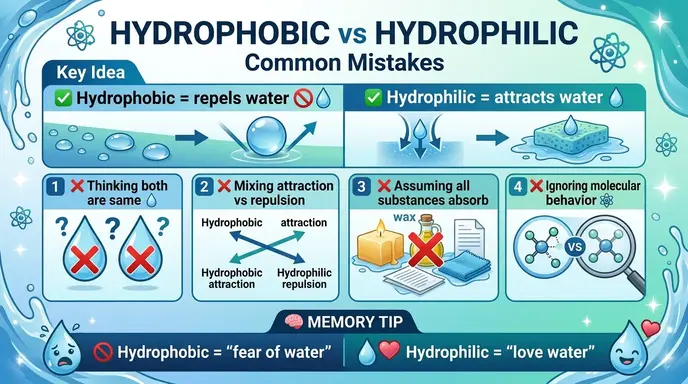
Confusing the Terms
❌ Hydrophobic means attracts water.
✅ Hydrophobic repels water.
Remember: “phobic” = fear.
Mixing Examples
❌ Oil is hydrophilic.
✅ Oil is hydrophobic.
Oil does not mix with water.
Ignoring Molecular Nature
❌ All substances behave the same with water.
✅ Molecular structure determines interaction.
Polarity matters.
Assuming Only Liquids Show This
❌ Only liquids can be hydrophobic or hydrophilic.
✅ Solids and surfaces can also show these properties.
Applies to many materials.
Thinking It’s Only Scientific
❌ These terms are only used in labs.
✅ They apply to everyday materials and products.
Real-world relevance is high.
🌍 Real Life Examples with Difference Between Hydrophobic and Hydrophilic
1️⃣ Clothing
Waterproof jackets repel water
2️⃣ Cleaning
Soap interacts with water and oil
3️⃣ Biology
Cell membranes have both properties
4️⃣ Surfaces
Glass attracts water, wax repels it
🎯 WHEN TO USE EACH
Use hydrophobic when:
✔ Describing water-repelling materials
✔ Talking about non-polar substances
Use hydrophilic when:
✔ Describing water-attracting materials
✔ Talking about soluble substances
🤔 WHY PEOPLE GET CONFUSED IN DIFFERENCE BETWEEN HYDROPHOBIC AND HYDROPHILIC
- Similar scientific terms
- Opposite meanings not obvious
- Lack of chemistry background
- Misleading examples
⚙️ How Science Systems Understand Hydrophobic vs Hydrophilic
Scientific systems classify materials based on molecular interaction:
- Hydrophobic = non-polar interaction
- Hydrophilic = polar interaction
👉 Helps in chemistry, biology, and material science.
🧑🔬 EXPERT INSIGHT
In real scenarios, the difference between hydrophobic and hydrophilic is fundamental to understanding how life and materials function.
Most beginners see it as a simple water interaction concept, but in advanced science, it explains how cell membranes form, how drugs dissolve, and how cleaning products work. This small concept has massive real-world impact.
👉 Key insight:
Water interaction defines behavior.
❓ FAQ:
❓ What is the difference between hydrophobic and hydrophilic?
Hydrophobic repels water, hydrophilic attracts water.
❓ What does hydrophobic mean?
It means water-repelling.
❓ What does hydrophilic mean?
It means water-attracting.
❓ Is oil hydrophobic?
Yes, oil repels water.
❓ Is salt hydrophilic?
Yes, it dissolves in water.
❓ Why do they behave differently?
Because of molecular polarity.
❓ Are these properties important?
Yes, in science and daily life.
❓ Can a substance be both?
Some materials have both parts (like soap).
🏁 Conclusion
The difference between hydrophobic and hydrophilic comes down to how substances interact with water. Hydrophobic materials repel water and do not mix with it, while hydrophilic materials attract water and dissolve easily.
Understanding this difference helps explain many everyday phenomena, from how soap cleans to how cells function. In simple terms, hydrophobic means water-repelling, and hydrophilic means water-attracting.
Once you understand this, many scientific and real-world concepts become much easier to grasp.
Discover More Articles:

I’m Owen Parker, and I write articles that focus on explaining the differences between common concepts, tools, and ideas. I enjoy simplifying complex information and turning it into clear comparisons that anyone can understand. My work often covers science , education, technology, and everyday knowledge topics.