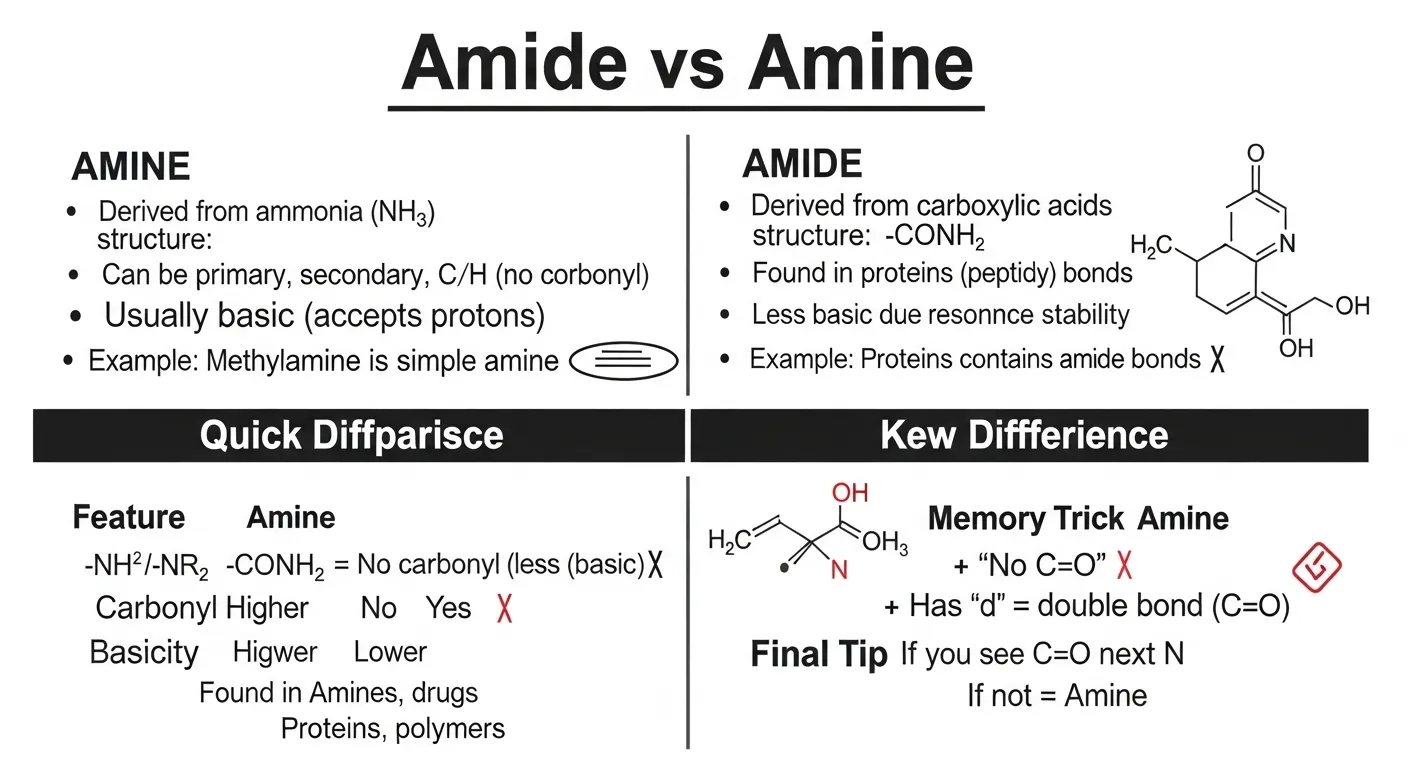
The difference between amide and amine is that amines are nitrogen-containing compounds derived from ammonia, while amides contain a carbonyl group attached to nitrogen.
Imagine a chemistry student learning organic compounds amines are simple structures formed by replacing hydrogen in ammonia. Amides, on the other hand, are more complex and are commonly found in proteins.
Understanding the difference between amide and amine helps in learning organic chemistry and biological systems.
Knowing the difference between amide and amine also improves understanding of chemical reactions and molecular structures.
🔑 Key Difference Between the Both
The main difference lies in structure and bonding.
- Amine: Contains nitrogen bonded to hydrogen or carbon.
- Amide: Contains nitrogen attached to a carbonyl group (C=O).
Examples:
- Amines are simpler compounds.
- They are derived from ammonia.
Examples:
- Amides are more complex.
- They are found in proteins.
🌍 Why Is Their Difference Necessary to Know?
Understanding the difference between amide and amine is important in chemistry, medicine, and biology. These compounds play key roles in chemical reactions and living organisms. Amines are involved in many industrial and biological processes, while amides form the backbone of proteins. Knowing the difference between amide and amine helps students and professionals understand molecular behavior, reactions, and applications in real life.
🔊 Pronunciation (US & UK)
| Word | US Pronunciation | UK Pronunciation |
| Amine | /ˈæmiːn/ | /ˈæmiːn/ |
| Amide | /ˈæmaɪd/ | /ˈæmaɪd/ |
➡️ Now let us explore the detailed difference between amide and amine step by step.
⚗️ Difference Between Amide and Amine
1️⃣ Structure
Amine has nitrogen bonded to hydrogen or carbon.
Amide has nitrogen attached to a carbonyl group.
Examples:
- Amine: R–NH₂ structure.
- It is simple.
Examples:
- Amide: R–CONH₂ structure.
- It includes C=O.
2️⃣ Functional Group
Amine contains an amino group (–NH₂).
Amide contains an amide group (–CONH₂).
Examples:
- Amines have nitrogen group.
- It is reactive.
Examples:
- Amides have carbonyl + nitrogen.
- It is stable.
3️⃣ Origin
Amine is derived from ammonia (NH₃).
Amide is derived from carboxylic acids.
Examples:
- Amines form by replacing hydrogen in ammonia.
- It is basic.
Examples:
- Amides form from acids and ammonia.
- It is different.
4️⃣ Basicity
Amine is basic.
Amide is less basic.
Examples:
- Amines accept protons.
- It reacts easily.
Examples:
- Amides are less reactive.
- It is stable.
5️⃣ Reactivity
Amine is more reactive.
Amide is more stable.
Examples:
- Amines react quickly.
- It forms bonds easily.
Examples:
- Amides resist reactions.
- It is stable due to resonance.
6️⃣ Bonding
Amine has single bonds.
Amide has resonance bonding.
Examples:
- Amines have simple bonds.
- It is flexible.
Examples:
- Amides have partial double bond.
- It is rigid.
7️⃣ Occurrence
Amine occurs in many chemicals.
Amide occurs in proteins.
Examples:
- Amines are in dyes and drugs.
- It is common.
Examples:
- Amides form peptide bonds.
- It is biological.
8️⃣ Polarity
Amine is moderately polar.
Amide is highly polar.
Examples:
- Amines dissolve in water.
- It is polar.
Examples:
- Amides are more polar.
- It forms strong bonds.
9️⃣ Hydrogen Bonding
Amine forms hydrogen bonds.
Amide forms stronger hydrogen bonds.
Examples:
- Amines form weak interactions.
- It is limited.
Examples:
- Amides form strong interactions.
- It is stable.
🔟 Usage
Amine is used in industry.
Amide is used in biology.
Examples:
- Amines are used in medicines.
- It is practical.
Examples:
- Amides are in proteins.
- It is essential.
🧠 Nature and Behaviour of Amide and Amine
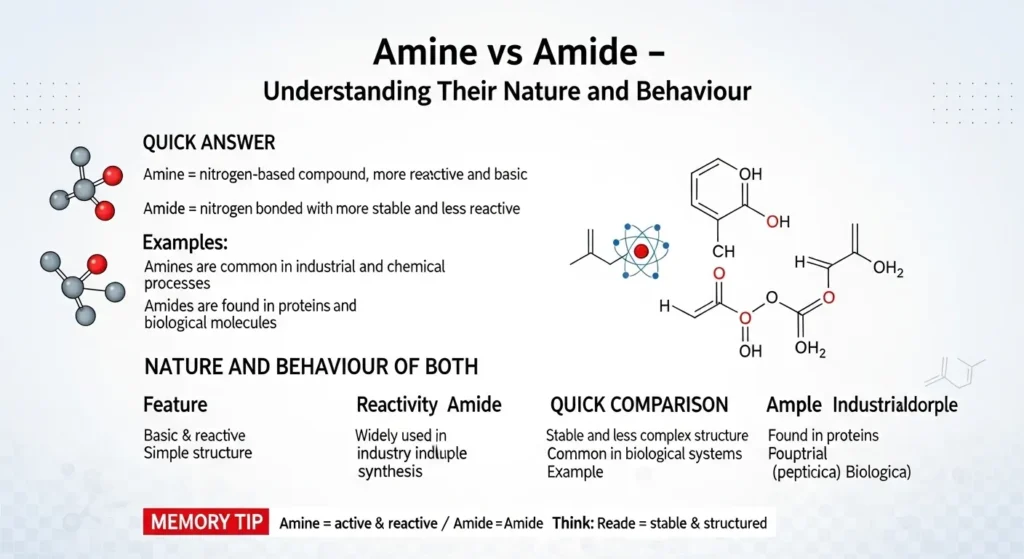
Amine
- Basic and reactive
- Simple structure
- Used in many industries
Amide
- Stable and less reactive
- Complex structure
- Found in biological systems
❓ Why People Are Confused About Their Use
People often confuse amide and amine because their names sound similar and both contain nitrogen. The confusion becomes clear when understanding their structure amines are simpler, while amides include a carbonyl group.
📋 Table: Difference and Similarity
| Feature | Amine | Amide |
| Structure | R–NH₂ | R–CONH₂ |
| Basicity | Strong | Weak |
| Reactivity | High | Low |
| Origin | Ammonia | Carboxylic acid |
| Occurrence | Chemicals | Proteins |
⚖️ Which Is Better in What Situation?
Amine
Amines are better in chemical reactions where reactivity is needed. They are useful in making drugs, dyes, and industrial products.
Amide
Amides are better for stability and biological functions. They are essential in proteins and play a key role in living organisms.
🎭 Use in Metaphors and Similes
Metaphor example:
- His ideas were like amines, quick and reactive.
Simile example:
- Her work was stable like an amide.
💬 Connotative Meaning
| Word | Connotation | Example |
| Amine | Activity, reactivity | His energy felt like amine. |
| Amide | Stability, strength | Her plan was like amide. |
🗣️ Expressions
Examples:
- Reactive like amine
- Stable like amide
📚 Works in Literature
- “Organic Chemistry” – Paula Yurkanis Bruice, 2016
- “Chemistry: The Central Science” – Brown, LeMay, 2018
🎬 Movies Related to Science
- “The Martian” – 2015, USA
- “Breaking Bad” – 2008, USA
❔ Frequently Asked Questions
1. What is the main difference between amide and amine?
Amines have nitrogen only, while amides have nitrogen with a carbonyl group.
2. Which is more reactive?
Amines are more reactive.
3. Which is found in proteins?
Amides are found in proteins.
4. Are both basic?
Amines are basic, amides are less basic.
5. Why are they similar?
Both contain nitrogen.
🌍 How Both Are Useful for Surroundings
Amines and amides are important in industry, medicine, and biology. They help in making products and understanding life processes.
✍️ Final Words for Both
Amine represents reactivity.
Amide represents stability.
Both are essential in chemistry.
🏁 Conclusion
Understanding the difference between amide and amine helps in learning organic chemistry more effectively. Amines are simple and reactive, while amides are stable and important in biological systems.
Although they may sound similar, their structures and roles are very different. Learning these differences improves knowledge of chemistry and its applications.
Discover More Articles:

Hi, I’m Nabeel Hassan. I love exploring interesting topics and explaining how different things compare with each other. Through my writing, I aim to provide clear, helpful, and easy to read explanations. I cover subjects related to learning, science, technology and general knowledge.